Why Typhoid Protection Matters
Typhoid fever is a potentially severe and life-threatening illness caused by Salmonella typhi, spread through contaminated food and water.3
It is common in parts of South Asia, Africa, Latin America, Southeast Asia, East Asia, and the Caribbean.2,4.
Please see Vivotif Prescribing Information .
Prevention and Protection
Antibiotic resistance is increasing, making prevention with vaccination even more important.5
Safe food and water practices are essential, but vaccination may provide added protection.1
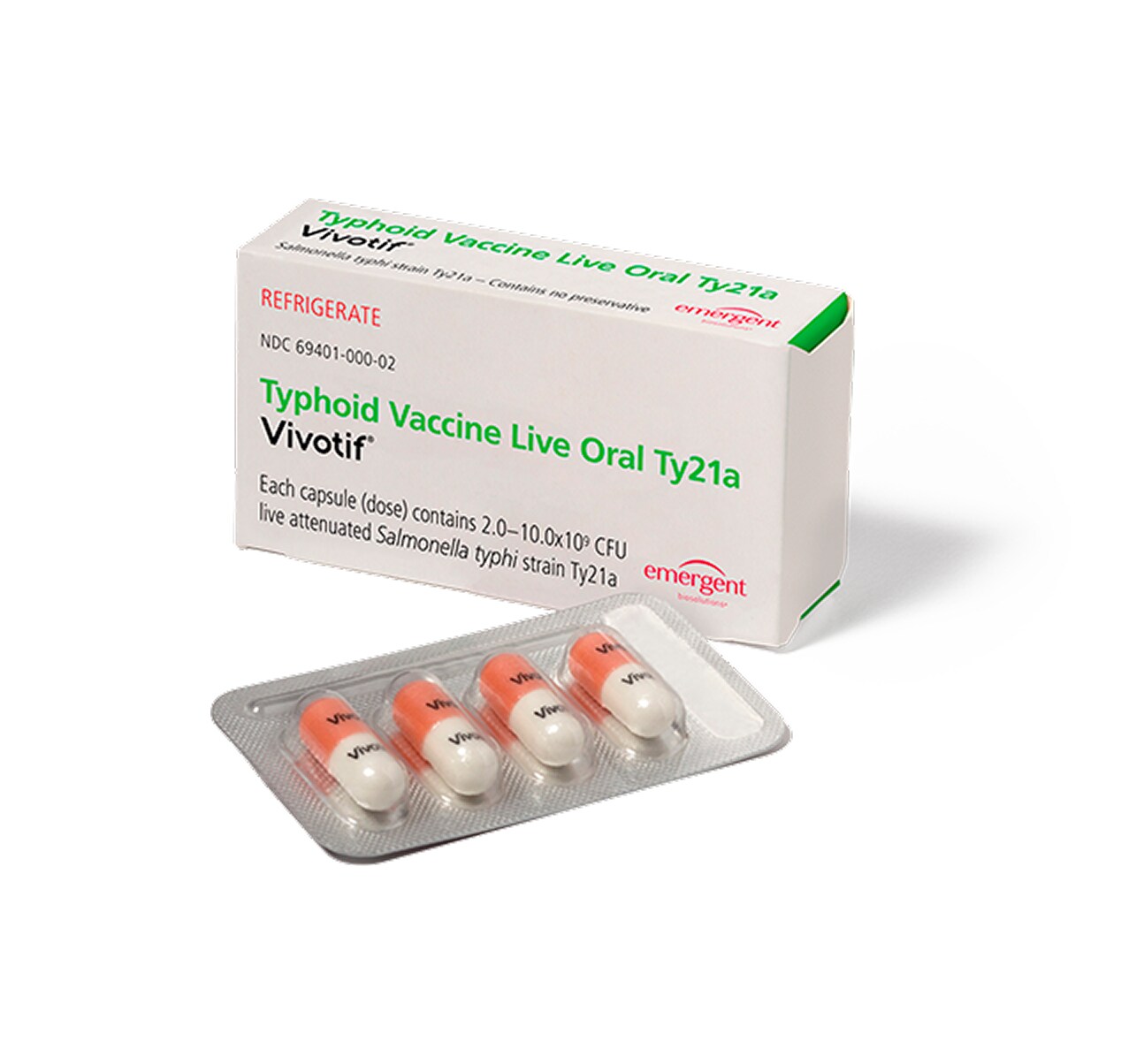
What is VIVOTIF?

The Only Oral Option
VIVOTIF is the only FDA-approved oral vaccine for typhoid fever.1,2

Approved for Ages 6+
VIVOTIF is for adults and children more than 6 years of age.1

Lasting Protection
VIVOTIF may help provide protection against typhoid fever for at least 5 years.1
VIVOTIF is recommended for people who are at a higher risk of getting typhoid fever.1This includes:
- People who travel to places where typhoid fever is more common
- People who have close contact with someone who has had typhoid fever or is known to be carrying the bacteria that causes it
- People who work in laboratories who could have contact with the bacteria that causes typhoid fever (Salmonella typhi)
One complete dosing schedule consists of 4 capsules.1
VIVOTIF is taken every other day for 7 days.1
All 4 doses of VIVOTIF should be taken at least 1 week before traveling.1
Swallow one capsule about 1 hour before a meal with a cold or lukewarm (temperature not to exceed body temperature, e.g., 37°C [98.6°F]) drink.1 It doesn’t matter what time you take your capsule, as long it’s at least 1 hour before a meal.
Swallow the capsule as soon after placing in the mouth as possible. Don't open or chew capsule.1
Do not freeze the capsules. Keep capsules refrigerated (35.6ºF–46.4ºF) until you're ready to take them.1
Text "TRAVEL" to 1-833-207-3377 on the day of your first dose to receive reminders.
All 4 doses of VIVOTIF should be taken at least 1 week before traveling.1
FAQ
- How long before my trip should I take VIVOTIF?
You should start taking VIVOTIF at least 2 weeks prior to your trip, so that you can complete your final dose at least 1 week before you plan to travel.1,6
- Who can use VIVOTIF?
VIVOTIF is for adults and children greater than 6 years of age.1 Immunization with VIVOTIF is not routinely recommended for people living in the United States1; however, VIVOTIF is recommended for people who are at a higher risk of getting typhoid fever. This includes1:
People who travel to places where typhoid fever is more common
People who have close contact with someone who has had typhoid fever or is known to be carrying the bacteria that causes it
People who work in laboratories who could have contact with the bacteria that causes typhoid fever (Salmonella typhi)
- What if I miss a dose of VIVOTIF?
It’s important to take VIVOTIF exactly as your healthcare provider or pharmacist tells you. VIVOTIF may not fully protect you from typhoid fever if you miss a dose.1 In case you miss a dose of VIVOTIF, contact your healthcare provider or pharmacist for instructions.
For full instructions on how to take VIVOTIF, see full Prescribing Information.
By accessing an external link you will be entering a third-party website, that is not operated and controlled by Walgreens. Walgreens does not take responsibility for the content or the privacy practices employed by other websites.
*Vaccines subject to availability. State-, age- and health-related restrictions may apply.
US-VIV-2500027 10/25
References:
- 1VIVOTIF (Typhoid Vaccine Live Oral Ty21a) [package insert]. Redwood City, CA: Emergent Travel Health Inc.
- 2CDC Yellow Book 2024: Typhoid & paratyphoid fever. Updated April 23, 2025. Accessed August 28, 2025. https://www.cdc.gov/yellow-book/hcp/travel-associated-infections-diseases/typhoid-and-paratyphoid-fever.html
- 3Centers for Disease Control and Prevention. About typhoid fever and paratyphoid fever. Updated April 25, 2024. Accessed August 28, 2025. https://www.cdc.gov/typhoid-fever/about
- 4Centers for Disease Control and Prevention. Preventing typhoid fever and paratyphoid fever. Updated August 5, 2025. Accessed August 28, 2025. https://www.cdc.gov/typhoid-fever/prevention/?CDC_AAref_Val=https://www.cdc.gov/typhoid-fever/prevention.html
- 5Centers for Disease Control and Prevention. Drug-resistant salmonella serotype typhi. Accessed August 28, 2025. https://www.cdc.gov/antimicrobial-resistance/media/pdfs/salmonella-typhi-508.pdf?CDC_AAref_Val=https://www.cdc.gov/drugresistance/pdf/threats-report/salmonella-typhi-508.pdf
- 6Centers for Disease Control and Prevention. Typhoid Vaccine VIS. Accessed August 28, 2024. https://www.cdc.gov/vaccines/hcp/current-vis/typhoid.html